Abstract
Review Article
Contrast-enhanced Susceptibility Weighted Imaging (CE-SWI) for the Characterization of Musculoskeletal Oncologic Pathology: A Pictorial Essay on the Initial Five-year Experience at a Cancer Institution
Raul F Valenzuela*, E Duran-Sierra, MA Canjirathinkal, B Amini, J Ma, KP Hwang, RJ Stafford, Keila E Torres, MA Zarzour, JA Livingston, JE Madewell, WA Murphy and CM Costelloe
Published: 02 April, 2024 | Volume 8 - Issue 1 | Pages: 030-045
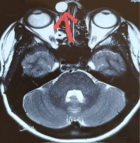
Susceptibility-weighted imaging (SWI) is based on a 3D high-spatial-resolution, velocity-corrected gradient-echo MRI sequence that uses magnitude and filtered-phase information to create images. It SWI uses tissue magnetic susceptibility differences to generate signal contrast that may arise from paramagnetic (hemosiderin), diamagnetic (minerals and calcifications) and ferromagnetic (metal) molecules. Distinguishing between calcification and blood products is possible through the filtered phase images, helping to visualize osteoblastic and osteolytic bone metastases or demonstrating calcifications and osteoid production in liposarcoma and osteosarcoma. When acquired in combination with the injection of an exogenous contrast agent, contrast-enhanced SWI (CE-SWI) can simultaneously detect the T2* susceptibility effect, T2 signal difference, contrast-induced T1 shortening, and out-of-phase fat and water chemical shift effect. Bone and soft tissue lesion SWI features have been described, including giant cell tumors in bone and synovial sarcomas in soft tissues. We expand on the appearance of benign soft-tissue lesions such as hemangioma, neurofibroma, pigmented villonodular synovitis, abscess, and hematoma. Most myxoid sarcomas demonstrate absent or just low-grade intra-tumoral hemorrhage at the baseline. CE-SWI shows superior differentiation between mature fibrotic T2* dark components and active enhancing T1 shortening components in desmoid fibromatosis. SWI has gained popularity in oncologic MSK imaging because of its sensitivity for displaying hemorrhage in soft tissue lesions, thereby helping to differentiate benign versus malignant soft tissue tumors. The ability to show the viable, enhancing portions of a soft tissue sarcoma separately from hemorrhagic/necrotic components also suggests its utility as a biomarker of tumor treatment response. It is essential to understand and appreciate the differences between spontaneous hemorrhage patterns in high-grade sarcomas and those occurring in the therapy-induced necrosis process in responding tumors. Ring-like hemosiderin SWI pattern is observed in successfully treated sarcomas. CE-SWI also demonstrates early promising results in separating the T2* blooming of healthy iron-loaded bone marrow from the T1-shortened enhancement in bone marrow that is displaced by the tumor.
SWI and CE-SWI in MSK oncology learning objectives:
SWI and CE-SWI can be used to identify calcifications on MRI.
Certain SWI and CE-SWI patterns can correlate with tumor histologic type.
CE-SWI can discriminate mature from immature components of desmoid tumors.
CE-SWI patterns can help to assess treatment response in soft tissue sarcomas.
Understanding CE-SWI patterns in post-surgical changes can also be useful in discriminating between residual and recurrent tumors with overlapping imaging features.
Read Full Article HTML DOI: 10.29328/journal.jro.1001062 Cite this Article Read Full Article PDF
Keywords:
Soft tissue sarcoma; Undifferentiated pleomorphic sarcoma; Multiparametric MRI; Treatment-induced hemorrhage; Susceptibility-weighted imaging
References
- Haacke EM, Xu Y, Cheng YC, Reichenbach JR. Susceptibility weighted imaging (SWI). Magn Reson Med. 2004 Sep;52(3):612-8. doi: 10.1002/mrm.20198. PMID: 15334582.
- Haacke EM, Mittal S, Wu Z, Neelavalli J, Cheng YC. Susceptibility-weighted imaging: technical aspects and clinical applications, part 1. AJNR Am J Neuroradiol. 2009 Jan;30(1):19-30. doi: 10.3174/ajnr.A1400. Epub 2008 Nov 27. PMID: 19039041; PMCID: PMC3805391.
- Böker SM, Adams LC, Bender YY, Fahlenkamp UL, Wagner M, Hamm B, Makowski MR. Differentiation of Predominantly Osteoblastic and Osteolytic Spine Metastases by Using Susceptibility-weighted MRI. Radiology. 2019 Jan;290(1):146-154. doi: 10.1148/radiol.2018172727. Epub 2018 Oct 30. PMID: 30375926.
- Rauscher A, Sedlacik J, Barth M, Mentzel HJ, Reichenbach JR. Magnetic susceptibility-weighted MR phase imaging of the human brain. AJNR Am J Neuroradiol. 2005; 26: 736-742. [PMID:15814914]
- Yamada N, Imakita S, Sakuma T, Takamiya M. Intracranial calcification on gradient-echo phase image: depiction of diamagnetic susceptibility. Radiology. 1996; 198: 171-178. [PMID: 8539373 DOI: 10.1148/radiology.198.1.8539373]
- Halefoglu AM, Yousem DM. Susceptibility weighted imaging: Clinical applications and future directions. World J Radiol. 2018 Apr 28;10(4):30-45. doi: 10.4329/wjr.v10.i4.30. PMID: 29849962; PMCID: PMC5971274.
- Sood S, Gupta R, Modi J, Sharma J. Susceptibility Weighted Imaging: Physics and Clinical Applications in Neuroimaging at 3 Tesla. European Congress of Radiology - ECR 2014. 2014. https://epos.myesr.org/poster/esr/ecr2014/C-1472
- Wu Z, Mittal S, Kish K, Yu Y, Hu J, Haacke EM. Identification of calcification with MRI using susceptibility-weighted imaging: a case study. J Magn Reson Imaging. 2009 Jan;29(1):177-82. doi: 10.1002/jmri.21617. PMID: 19097156; PMCID: PMC2646180.
- Martín-Noguerol T, Montesinos P, Casado-Verdugo OL, Beltrán LS, Luna A. Susceptibility Weighted Imaging for evaluation of musculoskeletal lesions. Eur J Radiol. 2021 May;138:109611. doi: 10.1016/j.ejrad.2021.109611. Epub 2021 Feb 28. PMID: 33677418.
- Barbosa JH, Santos AC, Salmon CE. Susceptibility weighted imaging: differentiating between calcification and hemosiderin. Radiol Bras. 2015 Mar-Apr;48(2):93-100. doi: 10.1590/0100-3984.2014.0010. PMID: 25987750; PMCID: PMC4433298.
- Valenzuela RF, Amini B, Duran-Sierra E, Canjirathinkal M, Idrees H, Madewell J, Costelloe C, Murphy W. Susceptibility-Weighted Imaging (SWI) in Benign and Malignant Tumors of the Musculoskeletal System. 2023. https://dx.doi.org/10.26044/ecr2023/C-19005
- Sierra DRF, Valenzuela M, Canjirathinkal J, Madewell W, Murphy C, Amini CB. Susceptibility Imaging (SWI) Morphologic Patterns and High-Order Radiomics of Baseline and Post-Treatment Advanced MRI of Extremity Soft Tissue Undifferentiated Pleomorphic Sarcoma.2023. https://dx.doi.org/10.26044/ecr2023/C-19060
- Jie L, Yong C, Xuemei B. Study of susceptibility-weighted imaging on MR and pathologic findings to distinguish benign or malignant soft tissue tumor. Chin J Oncol. 2017; 39(5).
- Takeuchi M, Matsuzaki K, Harada M. Clinical utility of susceptibility-weighted MR sequence for the evaluation of uterine sarcomas. Clin Imaging. 2019 Jan-Feb;53:143-150. doi: 10.1016/j.clinimag.2018.10.015. Epub 2018 Oct 13. PMID: 30340078.
- Nörenberg D, Ebersberger HU, Walter T, Ockert B, Knobloch G, Diederichs G, Hamm B, Makowski MR. Diagnosis of Calcific Tendonitis of the Rotator Cuff by Using Susceptibility-weighted MR Imaging. Radiology. 2016 Feb;278(2):475-84. doi: 10.1148/radiol.2015150034. Epub 2015 Sep 3. PMID: 26347995.
- Nörenberg D, Armbruster M, Bender YN, Walter T, Ebersberger HU, Diederichs G, Hamm B, Ockert B, Makowski MR. Diagnostic performance of susceptibility-weighted magnetic resonance imaging for the assessment of sub-coracoacromial spurs causing subacromial impingement syndrome. Eur Radiol. 2017 Mar;27(3):1286-1294. doi: 10.1007/s00330-016-4441-0. Epub 2016 Jun 10. PMID: 27287483.
- Bender YY, Diederichs G, Walter TC, Wagner M, Liebig T, Rickert M, Hermann KG, Hamm B, Makowski MR. Differentiation of Osteophytes and Disc Herniations in Spinal Radiculopathy Using Susceptibility-Weighted Magnetic Resonance Imaging. Invest Radiol. 2017 Feb;52(2):75-80. doi: 10.1097/RLI.0000000000000314. PMID: 27548342.
- Bender YY, Böker SM, Diederichs G, Walter T, Wagner M, Fallenberg E, Liebig T, Rickert M, Hamm B, Makowski MR. MRI for the detection of calcific features of vertebral haemangioma. Clin Radiol. 2017 Aug;72(8):692.e1-692.e7. doi: 10.1016/j.crad.2017.02.018. Epub 2017 Mar 19. PMID: 28330684.
- Ulas ST, Ziegeler K, Richter ST, Ohrndorf S, Poddubnyy D, Makowski MR, Diekhoff T. CT-like images in MRI improve specificity of erosion detection in patients with hand arthritis: a diagnostic accuracy study with CT as standard of reference. RMD Open. 2022 Feb;8(1):e002089. doi: 10.1136/rmdopen-2021-002089. PMID: 35177555; PMCID: PMC8860086.
- Akhavi Milani A, Daghighi MH, Mirza-Aghazadeh-Attari M, Jalili J, Mahmoudpour M, Daghighi S. The diagnostic value of susceptibility-weighted imaging for identifying acute intraarticular hemorrhages. Skeletal Radiol. 2022 Sep;51(9):1777-1785. doi: 10.1007/s00256-022-04016-6. Epub 2022 Feb 25. PMID: 35212784.
- Sieron DA, Drakopoulos D, Mitrakovic M, Tombarkiewicz M, Knap D, Platzek I, Tomalski M, Christe A. Assessment of 3-T MRI using susceptibility-weighted imaging to detect and evaluate intra- or periarticular blood metabolites and meniscal tears of the knee. Pol J Radiol. 2019 Sep 6;84:e340-e346. doi: 10.5114/pjr.2019.88480. PMID: 31969947; PMCID: PMC6964353.
- Böker SM, Adams LC, Bender YY, Wagner M, Diekhoff T, Fallenberg E, Hamm B, Makowski MR. Evaluation of vertebral body fractures using susceptibility-weighted magnetic resonance imaging. Eur Radiol. 2018 May;28(5):2228-2235. doi: 10.1007/s00330-017-5195-z. Epub 2017 Dec 19. PMID: 29260364.
- Liu J, Chen Y, Bao XM, Ling XL, Ding JP, Zhang ZK. [Study of susceptibility weighted imaging on MR and pathologic findings to distinguish benign or malignant soft tissue tumor]. Zhonghua Zhong Liu Za Zhi. 2017 May 23;39(5):350-354. Chinese. doi: 10.3760/cma.j.issn.0253-3766.2017.05.006. PMID: 28535651.
- Nishibori H, Kato H, Kawaguchi M, Nagano A, Matsuo M. T2*-weighted MR imaging findings of giant cell tumors of bone: radiological-pathological correlation. Jpn J Radiol. 2019 Jun;37(6):473-480. doi: 10.1007/s11604-019-00829-z. Epub 2019 Mar 11. PMID: 30859458.
- Dhillon M, Davies AM, Benham J, Evans N, Mangham DC, Grimer RJ. Calcific myonecrosis: a report of ten new cases with an emphasis on MR imaging. Eur Radiol. 2004 Nov;14(11):1974-9. doi: 10.1007/s00330-004-2368-3. Epub 2004 Oct 6. PMID: 15480695.
- Valenzuela RF, Madewell JE, Kundra V, Costelloe CM. Advanced Imaging in Musculoskeletal Oncology: Moving Away From RECIST and Embracing Advanced Bone and Soft Tissue Tumor Imaging (ABASTI)-Part II-Novel Functional Imaging Techniques. Semin Ultrasound CT MR. 2021 Apr;42(2):215-227. doi: 10.1053/j.sult.2020.08.013. Epub 2020 Aug 29. PMID: 33814107.
- Bush CH. The magnetic resonance imaging of musculoskeletal hemorrhage. Skeletal Radiol. 2000 Jan;29(1):1-9. doi: 10.1007/s002560050001. PMID: 10663582.
- Sparacia G, Speciale C, Banco A, Bencivinni F, Midiri M. Accuracy of SWI sequences compared to T2*-weighted gradient echo sequences in the detection of cerebral cavernous malformations in the familial form. Neuroradiol J. 2016 Oct;29(5):326-35. doi: 10.1177/1971400916665376. Epub 2016 Aug 22. PMID: 27549150; PMCID: PMC5033099.
- Hori M, Ishigame K, Kabasawa H. Pre- and postgadolinium enhanced susceptibility-weighted imaging at 1.5T for intracranial neoplasms: Contrast of pathologic lesions. Proc Intl Soc Mag Reson Med. 2007; 15:3764.
- Valenzuela RF, Amini B, Duran-Sierra E, Canjirathinkal MA, Madewell JE. Multiparametric MRI for the Assessment of Treatment Effect and Tumor Recurrence in Soft tissue sarcoma of the Extremities. J Radiol Oncol. 2023; 7: 058-065. DOI: 10.29328/journal.jro.1001055
- Adams LC, Bressem K, Böker SM, Bender YY, Nörenberg D, Hamm B, Makowski MR. Diagnostic performance of susceptibility-weighted magnetic resonance imaging for the detection of calcifications: A systematic review and meta-analysis. Sci Rep. 2017 Nov 14;7(1):15506. doi: 10.1038/s41598-017-15860-1. PMID: 29138506; PMCID: PMC5686169.
- Zulfiqar M, Dumrongpisutikul N, Intrapiromkul J, Yousem DM. Detection of intratumoral calcification in oligodendrogliomas by susceptibility-weighted MR imaging. AJNR Am J Neuroradiol. 2012 May;33(5):858-64. doi: 10.3174/ajnr.A2862. Epub 2012 Jan 19. PMID: 22268093; PMCID: PMC7968798.
- Child JR, Young CR, Amini B. Liposarcoma of the thigh with mixed calcification and ossification. Radiol Case Rep. 2016 Jul 1;11(3):217-21. doi: 10.1016/j.radcr.2016.05.010. PMID: 27594953; PMCID: PMC4996926.
- Berberat J, Grobholz R, Boxheimer L, Rogers S, Remonda L, Roelcke U. Differentiation between calcification and hemorrhage in brain tumors using susceptibility-weighted imaging: a pilot study. AJR Am J Roentgenol. 2014 Apr;202(4):847-50. doi: 10.2214/AJR.13.10745. PMID: 24660715.
- Wenaden AE, Szyszko TA, Saifuddin A. Imaging of periosteal reactions associated with focal lesions of bone. Clin Radiol. 2005 Apr;60(4):439-56. doi: 10.1016/j.crad.2004.08.017. PMID: 15767101.
- Ghosh PS, Ghosh D. Teaching neuroimages: MRI "target sign" and neurofibromatosis type 1. Neurology. 2012 Feb 28;78(9):e63. doi: 10.1212/WNL.0b013e318248df63. PMID: 22371419.
- Fox MG, Bancroft LW, Peterson JJ, Kransdorf MJ, Terkonda SP, O'Connor MI. MRI appearance of myocutaneous flaps commonly used in orthopedic reconstructive surgery. AJR Am J Roentgenol. 2006 Sep;187(3):800-6. doi: 10.2214/AJR.05.0484. PMID: 16928948.
- Knowles NG, Smith DL, Outwater EK. MRI diagnosis of brown tumor based on magnetic susceptibility. J Magn Reson Imaging. 2008 Sep;28(3):759-61. doi: 10.1002/jmri.21441. PMID: 18777537.
- Carvalho R, Kurochka S, Rocha J, Fernandes JS. Brown tumor of the mandible: Magnetic susceptibility demonstrated by MRI. Radiol Case Rep. 2015 Dec 7;7(2):662. doi: 10.2484/rcr.v7i2.662. PMID: 27326285; PMCID: PMC4899808.
- Valenzuela RF, Sierra ED, Canjirathinkal MA, Costelloe CM, Madewell JE, Murphy WA, Amini B. Early results in the novel use of contrast-enhanced susceptibility-weighted imaging in the assessment of response and progression in desmoid fibromatosis: A pilot study in a specialized cancer institution. Tumor Discovery. 2023; 2(3): 1414. https://doi.org/10.36922/td.1414
- Wardelmann E, Haas RL, Bovée JV, Terrier P, Lazar A, Messiou C, LePechoux C, Hartmann W, Collin F, Fisher C, Mechtersheimer G, DeiTos AP, Stacchiotti S, Jones RL, Gronchi A, Bonvalot S. Evaluation of response after neoadjuvant treatment in soft tissue sarcomas; the European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group (EORTC-STBSG) recommendations for pathological examination and reporting. Eur J Cancer. 2016 Jan;53:84-95. doi: 10.1016/j.ejca.2015.09.021. Epub 2015 Dec 14. PMID: 26700077.
Figures:
Similar Articles
-
Contrast-enhanced Susceptibility Weighted Imaging (CE-SWI) for the Characterization of Musculoskeletal Oncologic Pathology: A Pictorial Essay on the Initial Five-year Experience at a Cancer InstitutionRaul F Valenzuela*, E Duran-Sierra, MA Canjirathinkal, B Amini, J Ma, KP Hwang, RJ Stafford, Keila E Torres, MA Zarzour, JA Livingston, JE Madewell, WA Murphy, CM Costelloe. Contrast-enhanced Susceptibility Weighted Imaging (CE-SWI) for the Characterization of Musculoskeletal Oncologic Pathology: A Pictorial Essay on the Initial Five-year Experience at a Cancer Institution. . 2024 doi: 10.29328/journal.jro.1001062; 8: 030-045
-
Radiomics by Quantitative Diffusion-weighted MRI for Predicting Response in Patients with Extremity Soft-tissue Undifferentiated Pleomorphic SarcomaRF Valenzuela*, E Duran-Sierra, M Canjirathinkal, B Amini, KE Torres, RS Benjamin, J Ma, WL Wang, KP Hwang, RJ Stafford, C Wu, AM Zarzour, AJ Bishop, S Lo, JE Madewell, R Kumar, WA Murphy Jr, CM Costelloe. Radiomics by Quantitative Diffusion-weighted MRI for Predicting Response in Patients with Extremity Soft-tissue Undifferentiated Pleomorphic Sarcoma. . 2024 doi: 10.29328/journal.jro.1001066; 8: 064-071
Recently Viewed
-
Environmental Factors Affecting the Concentration of DNA in Blood and Saliva Stains: A ReviewDivya Khorwal*, GK Mathur, Umema Ahmed, SS Daga. Environmental Factors Affecting the Concentration of DNA in Blood and Saliva Stains: A Review. J Forensic Sci Res. 2024: doi: 10.29328/journal.jfsr.1001057; 8: 009-015
-
Markov Chains of Molecular Processes of Biochemical MaterialsOrchidea Maria Lecian*. Markov Chains of Molecular Processes of Biochemical Materials. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001076; 7: 001-005
-
Generation of Curved Spacetime in Quantum FieldSarfraj Khan*. Generation of Curved Spacetime in Quantum Field. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001077; 7: 006-009
-
Optimizing Milk Safety: Applying Nuclear Techniques in X-ray Fluorescence Spectroscopy for Heavy Metal Quantification in Powdered Milk Consumed in SenegalPapa Macoumba Faye*, Djicknack Dione, Oumar Ndiaye, Moussa Hamady SY, Nogaye Ndiaye, Alassane Traore, Ababacar Sadikhe Ndao. Optimizing Milk Safety: Applying Nuclear Techniques in X-ray Fluorescence Spectroscopy for Heavy Metal Quantification in Powdered Milk Consumed in Senegal. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001078; 7: 010-015
-
Thermoelectric Materials Based on Lead Telluride and Prospects for their Practical ApplicationYuriy Pavlovskyy*, Nadiya Pavlovska. Thermoelectric Materials Based on Lead Telluride and Prospects for their Practical Application. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001079; 7: 016-018
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."